No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
5HT Receptors
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
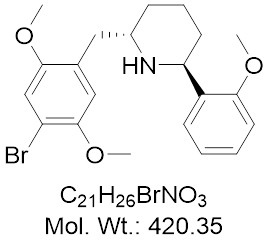
View larger Juncosamine (DMBMPP)
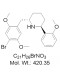
AOB11931
CAS 1391499-52-7
Chemical Name: DMBMPP; (2S,6S)-2-(2,5-Dimethoxy-4-bromobenzyl)-6-(2-methoxyphenyl)piperidine
1000 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $67.15 | Total: $335.75 |
| 1 | 10 | $56.88 | Total: $568.80 |
| 1 | 25 | $48.19 | Total: $1,204.75 |
| 1 | 50 | $41.08 | Total: $2,054.00 |
| 1 | 100 | $35.55 | Total: $3,555.00 |
Data sheet
| Molecular Formula | C21H26BrNO3 |
| Molecular Weight | 420.35 |
| CAS Numbers | 1391499-52-7 |
| Storage Condition | 0C Short Term -20C Long Term |
| Solubility | DMSO |
| Purity | 98% by HPLC |
| Synonym | DMBMPP; 5-HT2A receptor agonist-3 |
| IUPAC/Chemical Name | (2S,6S)-2-(2,5-Dimethoxy-4-bromobenzyl)-6-(2-methoxyphenyl)piperidine |
| InChl Key | KMVGLBONODPTDY-YJBOKZPZSA-N |
| InChl Code | COC1=C([C@H]2N[C@@H](CCC2)CC3=C(C=C(C(OC)=C3)Br)OC)C=CC=C1 |
| References | 1) Juncosa JI Jr, et al. Extensive rigid analogue design maps the binding conformation of potent N-benzylphenethylamine 5-HT2A serotonin receptor agonist ligands. ACS Chem Neurosci. 2013;4(1):96-109. |
More info
The most selective agonist for the human 5-HT2A receptor
What is DMBMPP?
DMBMPP (also known as juncosamine; chemical name: 2-(2,5-dimethoxy-4-bromobenzyl)-6-(2-methoxyphenyl) piperidine) is a synthetic compound. Wikipedia+2aobious.com+2
It belongs to a class of molecules derived from phenethylamines (specifically NBOMe-type derivatives) but incorporates a piperidine ring, making the molecule more rigid. Wikipedia+1
First described in the literature by a team including David E. Nichols (Purdue University) in ~2011. Wikipedia+1
Main functionality / pharmacology
DMBMPP is highly selective for the human serotonin receptor subtype 5‑HT₂A receptor (a G-protein-coupled receptor) and acts as an agonist at that receptor. Wikipedia+2PMC+2
The selectivity: The (S,S) isomer of DMBMPP has a Kₙᵢ (binding affinity) of ~2.5 nM at the human 5-HT₂A receptor, and ~124-fold selectivity over the similar 5-HT₂C receptor subtype. Wikipedia+1
Because 5-HT₂A is the receptor subtype most associated with classical psychedelic effects, DMBMPP is considered to have hallucinogen-like potential (based on animal models) albeit it is primarily a research tool compound. Wikipedia+1
Additional research suggests DMBMPP may also influence microtubule polymerization (i.e., cytoskeletal dynamics) in vitro, indicating possible off-target actions or broader cell-biological effects. Nature
Use / research context
DMBMPP is not primarily a therapeutic drug; rather it is used in research settings as a “tool compound” for studying 5-HT₂A receptor pharmacology because of its unusually high selectivity. PMC+1
One of the reasons for its limited wider use is that its chemical synthesis is relatively difficult / inaccessible, which limits its adoption as a widespread pharmacological agent. Wikipedia
Key limitations & caveats
Because it is so potent and selective, caution is needed in interpreting results: high selectivity does not always mean safe or predictable in vivo effects.
Human data are extremely limited or non-existent (at least that is publicly available) — so its effects in humans, its safe dosage, toxicity profile etc., are largely unknown.
The research suggests the possibility of microtubule / cytoskeleton interactions, which complicates the “pure receptor agonist” picture. Nature
Any compound with strong 5-HT₂A agonist activity has the potential for psychoactive/hallucinogenic effects, which brings regulatory and safety issues.
Summary
In short:
DMBMPP is a synthetic, highly selective agonist for the serotonin 5-HT₂A receptor, used in research to probe serotonin receptor function and related pharmacology. Its rigidity and design enable it to achieve one of the highest selectivities known for that receptor subtype. It is not a mainstream therapeutic drug and is mostly of interest in neuropharmacology / receptor biology / psychedelic-mechanism studies.