No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Allosteric
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
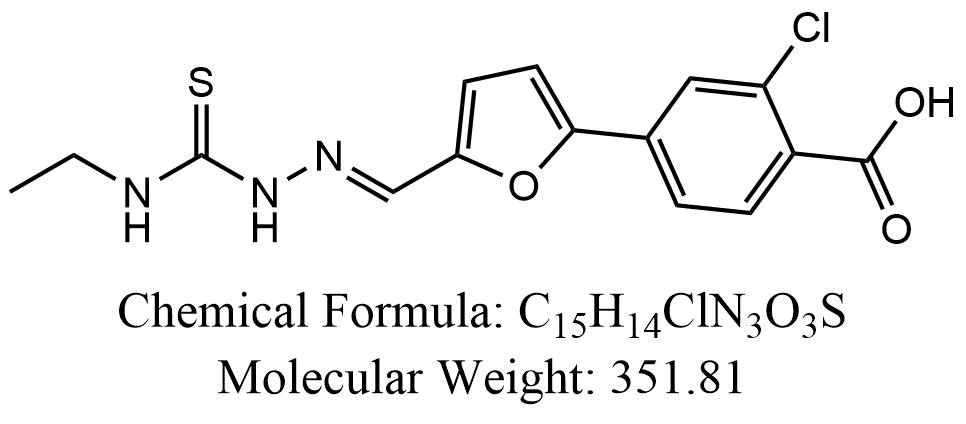 View larger
View larger PKUMDL-WQ-2201
AOBS8014
CAS No: 592474-91-4
Chemical Name: 2-Chloro-4-[5-[(ethylcarbamothioylhydrazinylidene)methyl]furan-2-yl]benzoic acid
1000 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $16.15 | Total: $80.75 |
| 1 | 10 | $13.68 | Total: $136.80 |
| 1 | 25 | $11.59 | Total: $289.75 |
| 1 | 50 | $9.88 | Total: $494.00 |
| 1 | 100 | $8.55 | Total: $855.00 |
Data sheet
| Molecular Formula | C15H14ClN3O3S |
| Molecular Weight | 351.81 |
| CAS Numbers | 592474-91-4 |
| Storage Condition | 0°C (short term), -20°C (long term), desiccated |
| Solubility | DMSA: 15 mg/mL |
| Purity | 98% by HPLC |
More info
Novel Selective Allosteric Inhibitor of PHGDH and Serine Synthesis with Anti-tumor Activity, binding to site II
What is PKUMDL-WQ-2201?
PKUMDL-WQ-2201 is a small molecule allosteric inhibitor of phosphoglycerate dehydrogenase (PHGDH). Forward 2000
More specifically, it is a negative allosteric modulator (i.e. it binds at a non-active site and downregulates PHGDH activity) Cell
In enzyme inhibition assays, its IC₅₀ for PHGDH is about 35.7 μM
Target & Pathway: PHGDH and Serine Biosynthesis
PHGDH (3-phosphoglycerate dehydrogenase) is the first enzyme in the de novo serine synthesis pathway, converting 3-phosphoglycerate (a glycolytic intermediate) to 3-phosphohydroxypyruvate. Cell+3ijbs.com+3PMC+3
Because serine is a precursor for many biosynthetic processes (nucleotides, proteins, lipids, one-carbon metabolism), many cancer cells, especially those with PHGDH overexpression or amplification, rely on serine synthesis for proliferation. ijbs.com+5PMC+5
By inhibiting PHGDH, PKUMDL-WQ-2201 suppresses de novo serine synthesis, which can limit the metabolic support for tumor cell growth. PMC+
Mechanism / Binding Mode
PKUMDL-WQ-2201 is non-competitive with respect to NAD⁺ and substrate (i.e. it does not bind in the active or cofactor pocket) — rather, it acts allosterically.
The inhibitor is proposed to bind to an “allosteric site II”, located in the substrate-binding domain, which influences domain movements necessary for catalysis. PMC+3Forward 2000 (Oxon) Ltd+3Cell+3
Structural modeling (or docking) suggests it forms hydrogen bonds with residues such as K57, T59, T56, stabilizing the enzyme in an inactive conformation by restricting motions of rigid domains required for catalysis. Cell+3ijbs.com+3Cell+3
Because of this binding, the enzyme is prevented from closing its catalytic conformation fully, thus reducing its activity. Cell+3
Biological / Cellular Effects
In cancer cell lines, PKUMDL-WQ-2201 shows dose-dependent suppression of cell viability in various models. Cell
In xenograft mouse tumor models, it has been observed to inhibit tumor growth when combined with cancer cells that are dependent on PHGDH.
In one recent study of breast cancer lines, PKUMDL-WQ-2201 inhibited viability of MCF-7 (ER+ cells) but had limited effect on MDA-MB-231 (a triple-negative line) under certain conditions, suggesting cell context dependency. PMC
The inhibitor effectively reduces the flux through serine synthesis, thereby interfering with downstream metabolic pathways tied to proliferation and survival. PMC+4Cell
Strengths, Limitations, & Considerations
Strengths / advantages:
As an allosteric inhibitor, PKUMDL-WQ-2201 avoids competition with high intracellular concentrations of cofactors like NAD⁺, which can hamper competitive inhibitors’ efficacy. PMC+2Cell+2
It offers a route to selectively target tumors with PHGDH overexpression or dependency on de novo serine synthesis, potentially sparing normal tissues that rely less on this pathway. Forward 2000 (Oxon) Ltd+4PMC+4ijbs.com+4
In vivo data (in mice) support its capacity to impair tumor growth, which is promising as a proof-of-concept. PMC
Limitations / challenges:
The IC₅₀ is moderate (≈ 35.7 µM), which is not especially potent for many drug development standards. PMC
Because serine can also be imported from extracellular sources or synthesized via alternative pathways, tumors might bypass inhibition by compensatory metabolic flux. PMC+3ijbs.com+3PMC+3
The context specificity means not all cancer cell types will respond uniformly. As seen in the MCF-7 vs MDA-MB-231 contrast, sensitivity varies. PMC
Potential toxicity or off-target effects are not yet fully characterized in humans or large animal models — systemic inhibition of serine production could affect non-tumor tissues.
Because it is a research compound (not clinical grade), its pharmacokinetics, bioavailability, stability, and safety in humans remain unproven.

