No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Antibacterial
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
View larger MMV371
AOB18144
CAS: 1080645-02-8
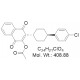
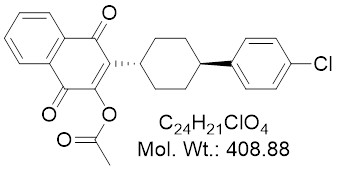
Chemical Name: mCBE161; 3-((1r,4r)-4-(4-Chlorophenyl)cyclohexyl)-1,4-dioxo-1,4-dihydronaphthalen-2-yl acetate
2500 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $73.95 | Total: $369.75 |
| 1 | 10 | $62.64 | Total: $626.40 |
| 1 | 25 | $53.07 | Total: $1,326.75 |
| 1 | 50 | $45.24 | Total: $2,262.00 |
| 1 | 100 | $39.15 | Total: $3,915.00 |
Data sheet
| Molecular Formula | C24H21ClO4 |
| Molecular Weight | 408.88 |
| CAS Numbers | 1080645-02-8 |
| Storage Condition | 0°C (short term), -20°C (long term), desiccated |
| Solubility | DMSO |
| Purity | 98% by HPLC |
| Synonym | MMV 371; MMV-371; mCBE161 |
| IUPAC/Chemical Name | 3-((1r,4r)-4-(4-Chlorophenyl)cyclohexyl)-1,4-dioxo-1,4-dihydronaphthalen-2-yl acetate |
| References | 1) Myriam El Gaaloul et. al.; Chemoprevention of malaria with long-acting oral and injectable drugs: an updated target product profile, Malaria Journal volume 23, Article number: 315 (2024) |
More info
MMV371 is a long-acting injectable (LAI) antimalarial drug developed by Medicines for Malaria Venture (MMV). It is designed to provide extended protection against malaria with a single intramuscular dose.
Mechanism of Action
MMV371 is a prodrug of atovaquone, a well-established antimalarial agent. By attaching an ester-linked acetyl group to atovaquone, MMV371 is designed to remain inactive until it is metabolized in the body. This modification allows for prolonged circulation and extended pharmacological activity. In preclinical studies, a single intramuscular injection of MMV371 maintained atovaquone levels above the minimum effective concentration for up to 69 days, compared to 21 days with atovaquone alone. Scripps Research
Target Parasites
MMV371 targets the mitochondrial cytochrome bc1 complex, inhibiting the electron transport chain in the malaria parasite. This action disrupts energy production, leading to parasite death. It is effective against both Plasmodium falciparum and Plasmodium vivax, the two most prevalent malaria parasites. mmv.org
Clinical Development
A Phase 1 clinical trial for MMV371 commenced in 2024 in Nottingham, UK. The trial aims to assess the drug's safety, tolerability, and pharmacokinetics in healthy volunteers. Pending successful outcomes, clinical trials in malaria-endemic regions are anticipated to begin in 2026. Scripps Research
Potential Benefits
Extended Protection: A single dose could offer up to three months of protection against malaria.
Simplified Dosing: Reduces the need for daily medication, improving patient compliance.
Asymptomatic Infection Clearance: Potential to clear asymptomatic malaria infections, reducing transmission.
Affordability: Designed to be cost-effective, making it accessible in resource-limited settings. Quotient Sciences