No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Autophagy
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
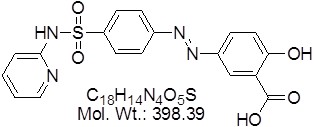
View larger Sulfasalazine
AOB0046
CAS 599-79-1
10000 Items
Molarity Calculation Cart®
HOW TO ORDER
Data sheet
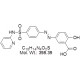
| Molecular Formula | C18H14N4O5S |
| Molecular Weight | 398.39 |
| CAS Numbers | 599-79-1 |
| Storage Condition | 0C Short Term, -20C Long Term |
| Solubility | DMSO |
| Purity | 98% by HPLC |
| Synonym | |
| IUPAC/Chemical Name | |
| InChl Key | |
| InChl Code | |
| SMILES Code | OC(=O)C1=CC(=CC=C1O)N=NC1=CC=C(C=C1)S(=O)(=O)NC1=CC=CC=N1 |
| References | Gadangi P, et al. J Immunol,1996, 156[5], 1937-1941. |
More info
Sulfasalazine (Azulfidine) is a synthetic salicylic acid derivative with affinity for elastin-containing connective tissues and formulated as a prodrug. Sulfasalazine induces iron death and inhibits NF-?B, TGF-? and COX-2.
1. Pharmacological Class
Type: Prodrug (inactive until metabolized)
Metabolized into:
Sulfapyridine – primarily responsible for antibacterial and some systemic effects
5-Aminosalicylic acid (5-ASA or mesalamine) – responsible for local anti-inflammatory effects in the gut
⚙️ 2. Mechanism of Action
Although the exact mechanism is complex and not fully understood, its main functional effects include:
a. Anti-inflammatory action
Inhibits cyclooxygenase (COX) and lipoxygenase pathways, reducing prostaglandin and leukotriene synthesis (key mediators of inflammation).
Scavenges reactive oxygen species (ROS), reducing oxidative tissue damage.
b. Immunomodulatory effects
Suppresses T-cell proliferation and cytokine release (e.g., TNF-α, IL-1, IL-6).
Inhibits NF-κB activation, a central transcription factor for inflammatory genes.
Reduces B-cell antibody production and natural killer (NK) cell activity.
c. Antibacterial effect (minor)
The sulfapyridine moiety has sulfonamide-type antibacterial activity, though not the main therapeutic goal.
3. Therapeutic Applications
| Disease / Condition | Functional Role of Sulfasalazine |
|---|---|
| Rheumatoid arthritis (RA) | Slows joint damage and inflammation; considered a first-line DMARD. |
| Ankylosing spondylitis | Reduces inflammation and stiffness, especially in peripheral joints. |
| Ulcerative colitis (UC) | Acts locally in the colon to reduce mucosal inflammation. |
| Crohn’s disease (colonic type) | Sometimes used, though less effective than in UC. |
| Psoriatic arthritis | Helps control synovial inflammation and pain. |
4. Cellular / Molecular Research Uses
In laboratory or experimental studies, sulfasalazine is often used to:
Inhibit NF-κB signaling in cell culture models.
Induce oxidative stress resistance or modulate glutathione metabolism.
Study cancer cell apoptosis and autophagy, since sulfasalazine inhibits xCT (SLC7A11), a cystine/glutamate antiporter, leading to glutathione depletion and ferroptosis in tumor cells.
⚠️ 5. Notable Considerations
Adverse effects: Nausea, rash, headache, reversible oligospermia, hematologic effects (e.g., agranulocytosis, anemia).
Monitoring: CBC and liver function during therapy.
Folate metabolism interference: Often supplemented with folic acid.