No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Epigenetics
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
View larger SNDX-5613
AT12943
CAS 2169919-21-3
Revumenib
1000 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $48.45 | Total: $242.25 |
| 1 | 10 | $41.04 | Total: $410.40 |
| 1 | 25 | $34.77 | Total: $869.25 |
| 1 | 50 | $29.64 | Total: $1,482.00 |
| 1 | 100 | $25.65 | Total: $2,565.00 |
Data sheet
| Molecular Formula | C32H47FN6O4S |
| Molecular Weight | 630.82 |
| CAS Numbers | 2169919-21-3 |
| Storage Condition | 0C Short Term, -20C Long Term |
| Solubility | DMSO |
| Purity | 98% by HPLC |
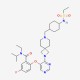
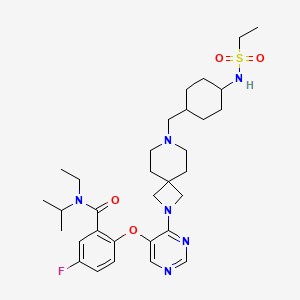
| SMILES Code | CCN(C(C)C)C(=O)c1cc(F)ccc1Oc1cncnc1N1CC2(C1)CCN(C[C@H]1CC[C@@H](CC1)NS(=O)(=O)CC)CC2 |
| References | A drug, SNDX-5613, to treat acute leukemia with a KMT2A translocation or an NPM1 mutation that has come back [relapsed] or has not gotten better with treatment [refractory]. |
More info
Revumenib represents a novel therapeutic approach targeting the menin–KMT2A interaction, offering a promising treatment option for patients with specific genetic alterations in acute leukemias. Its ability to modulate gene expression and promote leukemic cell differentiation underscores its potential as a targeted therapy in hematologic malignancies.Syndax+7Nature+7ASCOPubs+7
Mechanism of Action
Revumenib functions by disrupting the binding between menin and KMT2A, which is essential for the transcriptional activation of oncogenic genes such as HOX and MEIS1. These genes are implicated in maintaining the leukemogenic state of hematopoietic stem cells. By inhibiting this interaction, revumenib leads to the downregulation of these genes, promoting differentiation and apoptosis of leukemic cells .Nature+1
Clinical Applications
Revumenib is currently under investigation in clinical trials for the treatment of relapsed or refractory acute leukemias with KMT2A rearrangements or NPM1 mutations. The AUGMENT-101 trial has demonstrated promising results, with a 47% overall response rate and a 23% complete remission rate in patients with relapsed/refractory NPM1-mutant AML. Additionally, 64% of patients achieving complete remission were minimal residual disease-negative .Nature+2Wikipedia+2Wikipedia+4Targeted Oncology+4ScienceDirect+4