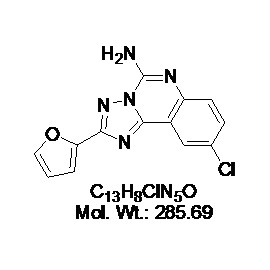
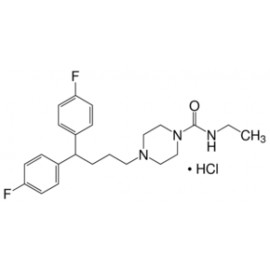
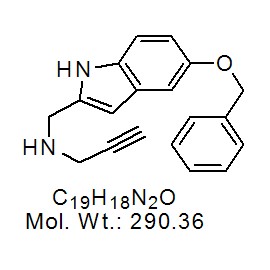
No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
G Protein-Coupled Receptor Ligands
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
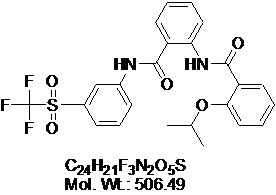 View larger
View larger RXFP1-Agonist-8
AOB4072
CAS No: 1482500-76-4
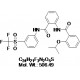
Chemical Name: 2-Isopropoxy-N-(2-(3-(trifluoromethylsulfonyl)phenylcarbamoyl)phenyl)benzamide; ML290; RXFP1-Agonist-8
915 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $14.45 | Total: $72.25 |
| 1 | 10 | $12.24 | Total: $122.40 |
| 1 | 25 | $10.37 | Total: $259.25 |
| 1 | 50 | $8.84 | Total: $442.00 |
| 1 | 100 | $7.65 | Total: $765.00 |
Data sheet
| Molecular Formula | C24H21F3N2O5S |
| Molecular Weight | 506.49 |
| CAS Numbers | 1482500-76-4 |
| Storage Condition | white powder at 4C for three years |
| Solubility | DMSO |
| Stock Solution Guide | Aliquot and Freeze (-20°C). Stock solutions are stable for up to 6 months at -20°C. |
| Synonym | ML290, ML-290, ML 290 |
| IUPAC/Chemical Name | 2-[[2-(1-methylethoxy)benzoyl]amino]-N-[3-[(trifluoromethyl)sulfonyl]phenyl]-benzamide |
| InChl Key | RSYHJSDOGMSLDH-UHFFFAOYSA-N |
| InChl Code | InChI=1S/C24H21F3N2O5S/c1-15(2)34-21-13-6-4-11-19(21)23(31)29-20-12-5-3-10-18(20)22(30)28-16-8-7-9-17(14-16)35(32,33)24(25,26)27/h3-15H,1-2H3,(H,28,30)(H,29,31) |
| SMILES Code | O=S(C1=CC=CC(NC(C2=CC=CC=C2NC(C3=C(OC(C)C)C=CC=C3)=O)=O)=C1)(C(F)(F)F)=O |
| References | 1) Xiao, J., et al. Discovery, optimization, and biological activity of the first potent and selective small-molecule agonist series of human relaxin receptor 1 (RXFP1). Probe Reports from the NIH Molecular Libraries Program (2012). 2) Diepenhorst, N.A.., et al. Investigation of interactions at the extracellular loops of the relaxin family peptide receptor 1 (RXFP1). J. Biol. Chem. 289(50), 34938-34952 (2014). 3) Huang, Z.,, et al. Activation of relaxin family receptor 1 from different mammalian species by relaxin peptide and small-molecule agonist ML290. Front. Endocrinol. (Lausanne) 6, 128 (2015). |
More info
The first potent and selective small-molecule agonist of human relaxin receptor 1 (RXFP1)
A cell-permeable 2-acetamido-N-phenylbenzamide that selectively activates human, but not mouse, LGR7/RXFP1-mediated cAMP induction (EC50 = 200 nM in THP1) via allosteric interaction with the ECL3 region without competing against ECL2-mediated relaxin binding or affecting AVPR1B- or LGR8/RXFP2-mediated cAMP induction. Although shown to be ~150-fold and 500-fold less potent than relaxin (RLX), respectively, in VEGF mRNA induction and cellular impedance assays, pharmacokinetic studies reveal superior in vivo stability to RLX and in vivo bioavailability in mice via oral (Cmax/Tmax = 604 nM/plasma/1 h and 1026 ng/g heart/1.5 h; 30 mg/kg) or intraperitoneal (Cmax/Tmax = 9.29 µM/plasma/1 h and 28.6 µmol/kg heart/1 h; 30 mg/kg) administration with good aqueous solubility (7 µM in PBS).
Reference:
1) Xiao, J., et al. 2013. Nat. Commun. 4, 1953.