No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Orphan Receptors
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
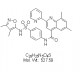
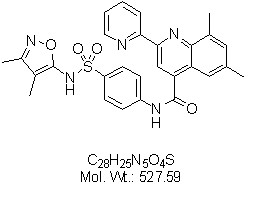
View larger ML193
AOB1576
CAS No: 713121-80-3
Chemical Name: N-{4-[(3,4-dimethyl-1,2-oxazol-5-yl)sulfamoyl]phenyl}-6,8-dimethyl-2-(pyridin-2-yl)quinoline-4-carboxamide; AC1LOE9L; CID-1261822
800 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $17.85 | Total: $89.25 |
| 1 | 10 | $15.12 | Total: $151.20 |
| 1 | 25 | $12.81 | Total: $320.25 |
| 1 | 50 | $10.92 | Total: $546.00 |
| 1 | 100 | $9.45 | Total: $945.00 |
Data sheet
| Molecular Formula | C28H25N5O4S |
| Molecular Weight | 527.59 |
| CAS Numbers | 713121-80-3 |
| Storage Condition | 0°C (short term), -20°C (long term), desiccated |
| Solubility | DMSO |
| Purity | 98% by HPLC |
| IUPAC/Chemical Name | N-[4-[[(3,4-dimethyl-5-isoxazolyl)amino]sulfonyl]phenyl]-6,8-dimethyl-2-(2-pyridinyl)-4-quinolinecarboxamide |
| InChl Key | HTSLEZOTMYUPLU-UHFFFAOYSA-N |
| InChl Code | InChI=1S/C28H25N5O4S/c1-16-13-17(2)26-22(14-16)23(15-25(31-26)24-7-5-6-12-29-24)27(34)30-20-8-10-21(11-9-20)38(35,36)33-28-18(3)19(4)32-37-28/h5-15,33H,1-4H3,(H,30,34) |
| SMILES Code | O=C(C1=C(C=C(C)C=C2C)C2=NC(C3=CC=CC=N3)=C1)NC4=CC=C(S(NC5=C(C)C(C)=NO5)(=O)=O)C=C4 |
| References | 1) Heynen-Genel, S., Dahl, R., Shi, S., et al. Screening for selective ligands for GPR55. NIH Molecular Libraries (2010). 2) Kotsikorou, E., Sharir, H., Shore, D.M., et al. Identification of the GPR55 antagonist binding site using a novel set of high-potency GPR55 selective ligands. Biochemistry 52(52), 9456-9469 (2013). |
More info
Selective Antagonists of GPR55
Functionality
Target:
ML-193 binds to GPR55, which is a G protein–coupled receptor. Discovery Dundee+1
GPR55 has been linked to a variety of physiological and pathological processes, including inflammatory pain, neuropathic pain, metabolism, bone development, and cancer.
Antagonist Activity:
Its IC₅₀ against GPR55 is ~ 221 nM, meaning it inhibits GPR55 signaling at that concentration.
It shows strong selectivity: for example, it’s >27-fold more selective for GPR55 over GPR35, CB1, and CB2 receptors.
Cellular Effects (In Vitro):
In U2OS cells, ML-193 inhibits β-arrestin trafficking that’s normally induced by the GPR55 agonist L-α-lysophosphatidylinositol (LPI).
It also reduces ERK1/2 phosphorylation (a downstream signaling event) in response to LPI, with an IC₅₀ of ~0.2 µM.
In human neural stem cells (hNSCs), ML-193 can dampen proliferation and reduce differentiation induced by GPR55 agonists.
In Vivo Effects:
In a rat model of Parkinson’s disease (using 6-hydroxydopamine lesioning), intra-striatal administration of ML-193 (1–5 µg per rat) improved motor coordination and reduced sensorimotor deficits.
This suggests potential neuroprotective or symptomatic benefits via blocking GPR55 in the brain.
Pharmacological Utility:
Because of its potency and selectivity, ML-193 is used as a research tool to probe GPR55 function. Karger Publishers
It helps scientists understand how GPR55 contributes to physiological processes and disease, by selectively blocking the receptor.
Why It's Important
Therapeutic Relevance: Since GPR55 is implicated in pain, cancer, and neurodegeneration, ML-193 (or derivatives) might help in the development of new therapies, or at least in understanding disease mechanisms.
Tool Compound: It’s very valuable for basic research, particularly for dissecting signaling pathways mediated by GPR55.