Categories
- PROTAC Linker
- Protein Control Ligand
- Pathway Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- SARS-CoV
TRPM4 Inhibitor NBA
AOB12909
CAS 667411-04-3 (Free Acid)
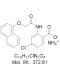
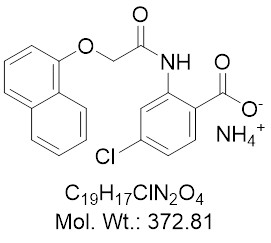
Chemical Name: 4-Chloro-2-(1-naphthyloxyacetamido)benzoic acid ammonium salt (NBA)
971 Items
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $24.65 | Total: $123.25 |
| 1 | 10 | $20.88 | Total: $208.80 |
| 1 | 25 | $17.69 | Total: $442.25 |
| 1 | 50 | $15.08 | Total: $754.00 |
| 1 | 100 | $13.05 | Total: $1,305.00 |
Overview
Novel specific TRPM4 inhibitor, blocking TRPM4 currents in HCT116 cells, reducing the viability of Colo205 cells. NBA is an ammonium salt form (more water soluble) of TRPM4-IN-2 (Free Acid).
Mechanism / Binding & SAR (Structure-Activity Relationships)
Binding Site / Mode of Inhibition
The precise binding site of NBA on TRPM4 has not yet been definitively mapped, although some recent work aims to identify it. PMC+1
One publication (“Identification of a binding site for small-molecule inhibitors …”) indicates that efforts are ongoing to pinpoint where anthranilic acid derivatives (like NBA) bind.
Because NBA is an aryloxyacyl-anthranilic acid derivative, its structural features (a naphthoxy substituent, anthranilic acid core) potentially allow interaction with hydrophobic pockets near the transmembrane domain, though this is speculative. Frontiers+2chimia.ch+2
Potency & Selectivity Compared to Other Inhibitors
NBA has an IC₅₀ of ~0.16 µM for TRPM4 inhibition.
In comparative studies with other TRPM4 inhibitors (e.g. CBA, LBA), NBA tends to show greater potency (i.e. require lower concentration for inhibition).
In colorectal cancer (CRC) cells (HCT116), NBA, CBA, and LBA were found to almost fully block TRPM4 currents (91.8–98.5%) in the low micromolar range; NBA was the most potent among them. MDPI+1
Some inhibition is non-reversible in CRC cells (i.e. washout does not fully restore the current), which suggests either very tight binding or possibly partial inactivation. MDPI
Functional Consequences of Inhibition / Downstream Effects
In CRC cell lines, inhibition of TRPM4 by NBA was associated with:
However, in some prostate cancer (PCa) cell models (DU145, LNCaP), NBA (and related inhibitors) did not reliably change functional readouts (viability, migration) despite blocking current—presumably because inhibition was incomplete or because TRPM4-mediated current alone is not entirely determinative of those phenotypes in those contexts. ScienceDirect+3MDPI+3PMC+3
Functional / Bioactive Profile
Target: TRPM4 (Transient Receptor Potential Melastatin 4) channel
Action: Inhibitor — it blocks or suppresses TRPM4 activity
Potency: IC₅₀ ≈ 0.16 µM (i.e. fairly potent)
In vitro evidence: At 50 µM, TRPM4-IN-2 can block ~88 ± 9% of endogenous TRPM4 currents in DU145 prostate cancer cells over a 0-500 s exposure period.
Research uses: It is used as a tool compound in studies of cancer (notably prostate cancer, colorectal cancer) to explore how TRPM4 influences tumor cell behavior.
Chemical Properties
| Molecular Formula | C19H17ClN2O4 |
| Molecular Weight | 372.81 |
| CAS Numbers | 667411-04-3 (free acid) |
| Solubility | DMSO and Water |
| Purity | 98% by HPLC |
| IUPAC/Chemical Name | 4-Chloro-2-(1-naphthyloxyacetamido)benzoic acid ammonium salt |
Storage and Handling
0°C (short term), -20°C (long term), desiccated
Synonyms
TRPM4-IN-2 Ammonium Salt