No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Sigma Receptors
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
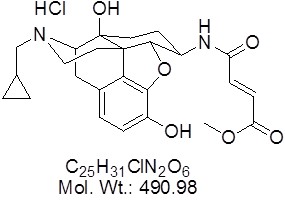
View larger beta-funaltrexamine Hydrochloride
AOB5999
CAS 72786-10-8
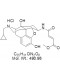
Chemical Name: ß-FNA hydrochloride, (E)-4-[[(5a,6ß)-17-(Cyclopropylmethyl)-4,5-epoxy-3,14-dihydroxymorphinan-6-yl]amino]-4-oxo-2-butenoic acid methyl ester hydrochloride
948 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $17.85 | Total: $89.25 |
| 1 | 10 | $15.12 | Total: $151.20 |
| 1 | 25 | $12.81 | Total: $320.25 |
| 1 | 50 | $10.92 | Total: $546.00 |
| 1 | 100 | $9.45 | Total: $945.00 |
Data sheet
| Molecular Formula | C25H31ClN2O6 |
| Molecular Weight | 490.98 |
| CAS Numbers | 72786-10-8 |
| Storage Condition | 0°C (short term), -20°C (long term), desiccated |
| Solubility | DMSO |
| Purity | 98% by HPLC |
| Synonym | ß-FNA hydrochloride |
| IUPAC/Chemical Name | (E)-4-[[(5a,6ß)-17-(Cyclopropylmethyl)-4,5-epoxy-3,14-dihydroxymorphinan-6-yl]amino]-4-oxo-2-butenoic acid methyl ester hydrochloride |
| InChl Key | BIPHUOBUKMPSQR-NQGXHZAGSA-N |
| InChl Code | 1S/C25H30N2O6.ClH/c1-32-20(30)7-6-19(29)26-16-8-9-25(31)18-12-15-4-5-17(28)22-21(15)24(25,23(16)33-22)10-11-27(18)13-14-2-3-14;/h4-7,14,16,18,23,28,31H,2-3,8-13H2,1H3,(H,26,29);1H/b7-6+;/t16-,18-,23+,24+,25-;/m1./s1 |
| SMILES Code | Cl.COC(=O)C=CC(=O)N[C@@H]1CC[C@@]2(O)[C@H]3Cc4ccc(O)c5OC1[C@]2(CCN3CC6CC6)c45 |
| References | 1) Jiang et al (1990) μ Antagonist and κ agonist properties of β-funaltrexamine (β-FNA) in vivo: long lasting spinal analgesia in mice. J.Pharmacol.Exp.Ther. 252 1006 PMID: 2156986 2) Takemorei et al (1981) The irreversible narcotic antagonist and reversible agonist properties of the fumarate methyl ester derivative of naltr. Eur.J.Pharmacol. 70 445 PMID: 6263637 |
More info
Selective irreversible µ opioid receptor antagonist, also being a kappa opioid receptor agonist
β-FNA hydrochloride (β-funaltrexamine hydrochloride) is a potent, irreversible μ-opioid receptor antagonist used in research. Here’s its main functionality:
Mechanism of action:
It binds selectively and irreversibly to μ-opioid receptors (MORs) through alkylation of a nucleophilic amino acid residue in the receptor binding pocket.
By doing so, it blocks receptor activation by agonists like morphine, fentanyl, or endogenous endorphins.
Selectivity:
Highly selective for MOR over δ- and κ-opioid receptors.
Often used in experiments to distinguish μ-mediated effects from other opioid receptor activities.
Applications in research:
Mapping μ-opioid receptor distribution and function.
Studying the role of μ-receptors in analgesia, reward pathways, and dependence.
Evaluating the effects of long-term receptor blockade (since it’s irreversible, recovery requires new receptor synthesis).
Here’s a clear side-by-side table comparing β-FNA hydrochloride, Naloxone, and Naltrexone:
| Feature | β-FNA hydrochloride | Naloxone | Naltrexone |
|---|---|---|---|
| Type | Irreversible μ-opioid receptor antagonist | Reversible competitive opioid antagonist | Reversible competitive opioid antagonist |
| Selectivity | Highly selective for μ (MOR) | μ > δ, κ | μ > δ, κ |
| Binding | Alkylates MOR → permanent inactivation | Binds competitively (reversible) | Binds competitively (reversible) |
| Duration of action | Very long (days, until new receptors are made) | Short (30–90 minutes) | Long (24–72 hours) |
| Clinical use | ❌ Research only | ✅ Emergency opioid overdose reversal | ✅ Maintenance therapy for opioid/alcohol dependence |
| Administration | Lab use (in vitro/in vivo animal studies) | IV, IM, intranasal | Oral tablets, IM depot injection |
| Purpose | Experimental tool for studying MOR function | Life-saving antidote for overdose | Long-term relapse prevention |