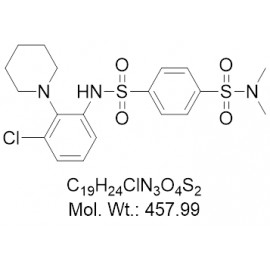
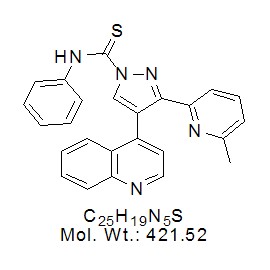
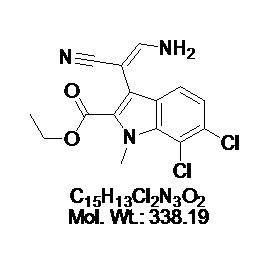
No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Enzyme Inhibitors
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
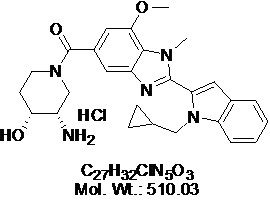 View larger
View larger GSK484
AOB6992
CAS No: 1652591-81-5
Chemical Name: ((3S,4R)-3-Amino-4-hydroxypiperidin-1-yl)(2-(1-(cyclopropylmethyl)-1H-indol-2-yl)-7-methoxy-1-methyl-1H-benzo[d]imidazol-5-yl)methanone hydrochloride
AOBIOUS launched this product in 2015
CITATIONS - Publications That Use AOBIOUS Product
512 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $24.65 | Total: $123.25 |
| 1 | 10 | $20.88 | Total: $208.80 |
| 1 | 25 | $17.69 | Total: $442.25 |
| 1 | 50 | $15.08 | Total: $754.00 |
| 1 | 100 | $13.05 | Total: $1,305.00 |
Data sheet
| Molecular Formula | C27H32ClN5O3 |
| Molecular Weight | 510.03 |
| CAS Numbers | 1652591-81-5 |
| Storage Condition | 0°C (short term), -20°C (long term), desiccated |
| Solubility | DMSO |
| Stock Solution Guide | 10 mMol/5.10 mg/1 mL; 5 mMol/2.55mg/1 mL in DMSO |
| Purity | 98% by HPLC |
| Synonym | GSK484; GSK-484; GSK 484. |
| IUPAC/Chemical Name | [(3S,4R)-3-amino-4-hydroxy-1-piperidinyl][2-[1-(cyclopropylmethyl)-1H-indol-2-yl]-7-methoxy-1-methyl-1H-benzimidazol-5-yl]-methanone |
| InChl Key | MULKOGJHUZTANI-ADMBKAPUSA-N |
| InChl Code | InChI=1S/C27H31N5O3.ClH/c1-30-25-20(11-18(13-24(25)35-2)27(34)31-10-9-23(33)19(28)15-31)29-26(30)22-12-17-5-3-4-6-21(17)32(22)14-16-7-8-16;/h3-6,11-13,16,19,23,33H,7-10,14-15,28H2,1-2H3;1H/t19-,23+;/m0./s1 |
| SMILES Code | O[C@@H]1CCN(C(C2=CC(N=C(C3=CC(C=CC=C4)=C4N3CC5CC5)N6C)=C6C(OC)=C2)=O)C[C@@H]1N.Cl |
| References | 1) Jones, J.E., Causey, C.P., Knuckley, B., et al. Protein arginine deiminase 4 (PAD4): Current understanding and future therapeutic potential. Curr. Opin. Drug Discov. Devel. 12(5), 616-627 (2009). 2) Lewis, H.D., Liddle, J., Coote, J.E., et al. Inhibition of PAD4 activity is sufficient to disrupt mouse and human NET formation. Nat. Chem. Biol. 11(3), 189-191 (2015). |
More info
GSK484 is a potent, well-characterized PAD4 inhibitor used extensively in research to dissect PAD4’s role in inflammation, immunity, cancer biology, and epigenetic regulation. Its reversible and selective nature, along with strong structural validation, make it an excellent tool—especially for cellular systems.
It selectively targets PAD4 over PAD1–3, disrupting citrullination—the conversion of arginine to citrulline—particularly important in neutrophil function and inflammation WikipediaAbcamStructural Genomics Consortium.
It binds to the low-calcium form of PAD4 with nanomolar potency (IC₅₀ ≈ 50 nM, increasing to ~250 nM in the presence of calcium) Structural Genomics Consortium
Structural studies revealed that GSK484 acts by inducing a conformational change—forming a β-hairpin in the PAD4 active site—distinct from older irreversible amidine inhibitors Structural Genomics Consortium.
Biological & Functional Effects
Inhibition of NETosis (Neutrophil Extracellular Trap formation): GSK484 blocks citrullination in neutrophils and prevents NET formation in both mouse and human cells, a process linked to thrombosis, autoimmune conditions, and cancer-related inflammation Structural Genomics ConsortiumAb
Anti-inflammatory and Radiosensitizing Effects:
In colorectal cancer models, GSK484 enhances radiosensitivity, promoting DNA double-strand breaks and cell death, while inhibiting NET formation PubMedWiley Online Library.
It has demonstrated anti-inflammatory benefits in other in vivo models, such as alleviating inflammation in giant cell myocarditis by suppressing NETosis ScienceDirect.
A Chemical Probe with Strong Selectivity
The Structural Genomics Consortium (SGC) and GlaxoSmithKline developed GSK484 as part of an epigenetic probe collection—making it a high-quality tool for studying PAD4’s biological role Structural Genomics ConsortiumIUPHAR/BPS Guide to Pharmacology.
The Chemical Probes Portal rates GSK484 as a mixed-mode PAD4 inhibitor with strong selectivity, recommending cellular use up to ~10 µM, but cautioning about calcium-dependent variability and calling attention to use of an inactive control (GSK106) in studies Chemical Probes Portal.