No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Ubiquitin-Activating Enzyme
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
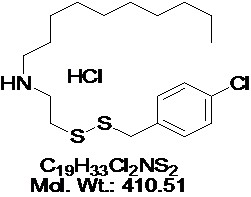
View larger NSC-624206
AOB3010
CAS No: 13116-77-3
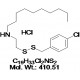
Chemical Name: NSC624206; N-[2-[(4-Chlorophenyl)methyldisulfanyl]ethyl]decan-1-amine Hydrochloride
498 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $15.30 | Total: $76.50 |
| 1 | 10 | $12.96 | Total: $129.60 |
| 1 | 25 | $10.98 | Total: $274.50 |
| 1 | 50 | $9.36 | Total: $468.00 |
| 1 | 100 | $8.10 | Total: $810.00 |
Data sheet
| Molecular Formula | C19H33Cl2NS2 |
| Molecular Weight | 410.51 |
| CAS Numbers | 13116-77-3 |
More info
NSC 624206 (CAS No. 13116-77-3) is a selective inhibitor of the ubiquitin-activating enzyme (E1), specifically targeting the UBA1 enzyme. This enzyme catalyzes the initial step in the ubiquitin-proteasome system, a crucial process for regulating protein degradation and turnover.
Target and Mechanism of Action
Primary Target: Ubiquitin-activating enzyme (UBA1/E1)
Mechanism:
NSC 624206 inhibits the formation of the ubiquitin-thioester bond during ubiquitination.
Importantly, it does not inhibit the adenylation step of ubiquitin activation.
Effect: By blocking UBA1 activity, it prevents the ubiquitination of substrate proteins, leading to altered protein turnover in cells.
Biological Effects
p27 Stabilization:
p27, a tumor suppressor protein, is normally degraded via the ubiquitin-proteasome system.
NSC 624206 prevents p27 ubiquitination, resulting in accumulation of p27, which can inhibit cell cycle progression.
IC₅₀: ~9 µM for UBA1 inhibition in vitro.
Cellular Effects:
Inhibition of UBA1 can modulate proteostasis, potentially inducing cell cycle arrest in cancer cell lines.
May affect other proteins regulated by ubiquitination, though specificity is primarily toward E1.
Research Applications
Used in cancer biology studies to investigate the role of ubiquitination in tumor suppressor regulation.
Tool compound for studying protein degradation pathways and ubiquitin-dependent signaling.
Can help in screening or validation of other inhibitors targeting the ubiquitin-proteasome system.
Structural Insights
Crystallographic studies have revealed that NSC 624206 binds to a specific site on the UBA1 enzyme, providing insights into its inhibitory mechanism. These structural details are valuable for designing more potent and selective inhibitors targeting the ubiquitin-activating enzyme. RSC PublishingBiocrick+3ScienceDirect+3ScienceDirect+3