No products
Product successfully added to your shopping cart
There are 0 items in your cart. There is 1 item in your cart.
Enzyme Inhibitors
- Protein Control Ligand
- Pathway Inhibitors
- Enzyme Inhibitors
- Kinase Inhibitors
- Protease
- Synthase
- p18
- p38
- p53
- p70
- p90
- Peptidase
- Carboxyl and Decarboxylases
- Ceramide Turnover Enzymes
- Chromatin Modifying Enzymes
- Cyclic Nucleotide Turnover Enzymes
- Glycerophospholipid Turnover Enzymes
- Hydroxylases
- Ubiquitin-Activating Enzyme
- Adenosine Deaminase
- Clathrin
- Nuclease
- p68
- ACE
- COX
- DHFR
- Neprilysin
- NF-κB
- RAF
- RAS
- Reductase
- ROR
- Topoisomerase
- Transferase
- Protein Inhibitors
- Transporter Inhibitors
- Cell Inhibition
- Synthase
- Receptor Tyrosine Phosphatases (RTP)
- AChE
- Peptidase
- Autophagy
- Toll-Like Receptor (TLR)
- Enzyme Inhibitors
- Function Modulators
- Activators
- G Protein-Coupled Receptor Ligands
- 5HT Receptors
- Adrenoceptor
- Angiotensin Receptor
- Cannabinoid Receptors
- CCK Receptors
- DA Receptors
- EAA Receptors
- Ghrelin Receptors
- GABA Receptors
- Histamine Receptors
- Leukotriene Receptors
- Metabotropic Glutamate Receptors
- Motilin Receptors
- Muscarinic Receptor
- Neuropeptide Receptors
- Opioid Receptors
- Orexin Receptors
- Orphan Receptors
- Prostanoid Receptors
- Proteinase-Activated Receptors
- Purinergic Receptors
- Ryanodine receptor
- Sigma Receptors
- Thrombin Receptor
- Vaniloid Receptor
- VIP and PACAP Receptors
- Neurotensin Receptors
- Urotensin Receptor
- Imidazoline receptor
- SMO Receptors
- Apelin Receptor
- β-arrestin/β2-adaptin
- KDM4
- Glucocorticoid Receptor
- Laminin Receptor
- AHR
- Amylin Receptor
- Bombesin Receptor
- Bradykinin Receptor
- CFTR
- CGRP Receptor
- CRFR
- Endothelin Receptor
- Ephrin Receptor
- Farnesoid X receptor (FXR)
- Glucagon Receptor
- Nuclear Receptor Ligands
- GDNF Receptors
- TNF Receptors
- Transcription Factors
- Chemokines
- Cytokine Receptors
- Biomarkers and Buffer Solutions
- Molecular Probes
- Stem Cell Research
- Alzheimer's Disease
- Apoptosis
- Cancer Research
- Epigenetics
- Metabolites
- PET/SPECT Imaging Precursors
- Customized Screening Library
- Ultra Pure Pharmacological Standard
- Tissue Microarray (TMA)
- Proteins and Antibodies
- Primary Cells
- ELISA KIT
- Natural Products
- Lab Equipments
- Humanized Mice for PDX Platform
- Rare Chemicals
- Custom Synthesis
- Antibacterial
- Antifungal
- Antioxidant
- Antiviral
- Molecular Glues
- PROTAC Linker
- SARS-CoV
 View larger
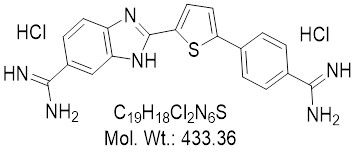
View larger DB818
AOB17565
CAS: 790241-43-9
Chemical Name: 2-(5-(4-Carbamimidoylphenyl)thiophen-2-yl)-1H-benzo[d]imidazole-6-carboximidamide dihydrochloride
835 Items
Molarity Calculation Cart®
HOW TO ORDER
Quantity Discount Table - Order More To Get More Price Discount
| Quantity | mg | Unit Price ($/mg or $/Unit) | Final Price |
|---|---|---|---|
| 1 | 5 | $16.15 | Total: $80.75 |
| 1 | 10 | $13.68 | Total: $136.80 |
| 1 | 25 | $11.59 | Total: $289.75 |
| 1 | 50 | $9.88 | Total: $494.00 |
| 1 | 100 | $8.55 | Total: $855.00 |
Data sheet
| Molecular Formula | C19H18Cl2N6S |
| Molecular Weight | 433.36 |
| CAS Numbers | 790241-43-9 |
| Storage Condition | 0°C (short term), -20°C (long term), desiccated |
| Solubility | DMSO |
| Purity | 98% by HPLC |
| IUPAC/Chemical Name | 2-(5-(4-Carbamimidoylphenyl)thiophen-2-yl)-1H-benzo[d]imidazole-6-carboximidamide dihydrochloride |
| References | 1) Depauw S, et al. J Med Chem. 2019 Jan 15 |
More info
Novel HOXA9 Transcription Factor Inhibitor, targeting the DNA minor groove, bindjng rapidly and specifically to parasite kinetoplast DNA (k-DNA)
What DB818 is
DB818 is a small‑molecule inhibitor that targets the interaction between the transcription factor HOXA9 and DNA.
Specifically, DB818 binds in the DNA minor groove at the HOXA9‑cognate binding sequence, thereby interfering with HOXA9’s ability to regulate its target genes.
It is intended for research use only (not an approved therapeutic) and is studied in the context of hematologic malignancies (particularly acute myeloid leukemia, AML) in which HOXA9 is dysregulated. PubMed+1
How DB818 works (mechanism of action)
HOXA9 is a homeobox‐domain transcription factor that plays a key role in normal hematopoietic development and is often overexpressed or aberrantly active in AML. Its overactivity is associated with poor prognosis. PubMed+1
DB818 disrupts the HOXA9/DNA binding interaction: one supplier reports a Kd_dd (dissociation constant) of ~4.6 nM for DB818 binding to a HOXA9‑cognate DNA sequence.
By blocking HOXA9’s binding, DB818 decreases the expression of HOXA9 target genes (such as MYB, MYC, BCL2) and increases expression of differentiation‐associated genes (e.g., FOS) in AML cells treated with the compound. PubMed+1
In functional terms, the treated AML cells show reduced proliferation, increased apoptosis (cell death), and signs of differentiation (i.e., release of the block on maturation) upon DB818 treatment. IIAR Journals+1
Research findings & key data
In a 2021 paper (Sonoda et al.), treatment of AML cell lines (OCI/AML3, MV4‑11, THP‑1) with DB818 led to suppressed growth, induced apoptosis, and down‑regulated HOXA9 target genes. PubMed
A 2024 study (Lambert et al.) reported that DB818 (alongside another compound DB1055) induced differentiation of HOXA9‑dependent AML cells, reduced leukemia burden in in vivo mouse models, and importantly had limited effect on normal CD34+ hematopoietic progenitor cells (suggesting some selectivity). PubMed
Practical/Research Considerations
Because DB818 acts by interfering with a transcription factor’s DNA binding, it falls under the category of “transcription factor inhibitors” — which are typically more challenging to develop than enzyme inhibitors.
Being research grade, use is restricted to lab/bench studies (cell‑based, animal models), not human clinical use.
Key controls when using DB818 in experiments:
Use appropriate HOXA9‐high vs HOXA9‐low cell lines to assess specificity.
Monitor target gene expression changes (MYB, BCL2, etc) and phenotypic readouts (proliferation, apoptosis, differentiation markers).
Consider off‑target effects: for example the Sonoda study noted among OCI/AML3 cells that MYC expression was not altered in the same way after HOXA9 knockdown as it was after DB818 treatment, indicating DB818 may have effects beyond just HOXA9. PubMed
Because the mechanism involves DNA‐binding and potentially non‑specific effects on DNA minor groove, dose, solvent, and formulation may be critical to interpretation.
Safety/toxicity in humans is unknown; all data so far are pre‑clinical.
Summary statement
In summary: DB818 is a laboratory‐grade inhibitor of HOXA9/DNA binding, developed to probe and potentially target HOXA9‑driven AML. It works by binding to the DNA minor groove at HOXA9 recognition sites, disrupting HOXA9 transcriptional regulation. This results in reduced proliferation, induced apoptosis, and differentiation of AML cells that depend on HOXA9. While promising in pre‑clinical models, it remains a research tool rather than a therapeutic agent.